Neuropharmacology & Neurotherapeutics
Upadacitinib
May. 14, 2026
MedLink, LLC
3525 Del Mar Heights Rd, Ste 304
San Diego, CA 92130-2122
Toll Free (U.S. + Canada): 800-452-2400
US Number: +1-619-640-4660
Support: service@medlink.com
Editor: editor@medlink.com
ISSN: 2831-9125
Toll Free (U.S. + Canada): 800-452-2400
US Number: +1-619-640-4660
Support: service@medlink.com
Editor: editor@medlink.com
ISSN: 2831-9125
Worddefinition
At vero eos et accusamus et iusto odio dignissimos ducimus qui blanditiis praesentium voluptatum deleniti atque corrupti quos dolores et quas.
This article describes the neurologic effects of various chemical agents used for chemical warfare or terrorism. The pathogenesis and pathophysiology as well as clinical manifestations of the important agents are described. Management is focused on organophosphates as chemical agents, particularly sarin. Neurologic manifestations can be acute, and there are some late sequelae in survivors.
The history of chemical weapons spans many centuries, but most important events occurred since World War I (16).
Abbreviations: CW=chemical weapons; CWC=Chemical Weapons Convention; OPCW=Organization for the Prohibition of Chemical Weapons, the international watchdog responsible for overseeing the implementation of the CWC. (Source: Caval...
Early use of chemical weapons. Aboriginal peoples on several continents employed toxins for hunting and warfare (eg, curare and amphibian-derived toxins as arrow poisons by aboriginal South Americans; "upas" as an arrow poison by Malaysians) (21).
The latex of the upas tree (Antiaris toxicari) contains intensely toxic cardenolides, including a cardiac glycoside named antiarin that is used as a toxin for arrows, darts, and blowdarts in Southeast Asian cultures. T...
However, many of the claims concerning the use of chemical weapons in ancient history are not supported by rigorous evidence and, although seemingly referred to in the introductions to numerous articles, editorials, and monographs on chemical weapons and terrorism, are unreferenced and uncritical rehashings of speculative and dubious claims, are often reiterated and amplified on social media (31). For example, oft-repeated claims of Assyrians poisoning enemy water supplies with ergot are unsupported by any rigorous evidence, even though it is known that Assyrians knew of ergot.
Much more plausible are accounts of the use of noxious smoke to incapacitate or sometimes to directly kill opponents, as employed, for example, by the Spartans lighting a mixture of wood, pitch, and sulfur under the walls of a besieged Athenian city during the Peloponnesian War (431-404 BCE). Another example is the Sassanians (Neo-Persians) burning bitumen and sulfur crystals to give off dense clouds of choking sulfur dioxide gas during the Roman-Persian Wars (54 BCE to 628 CE).
Napoleonic Wars (1803-1815). Thomas Cochrane (1775-1860), 10th Earl of Dundonald, styled "Lord Cochrane," was a British naval flag officer of the Royal Navy, mercenary, and radical politician. He had such success as a captain during the Napoleonic Wars that Napoleon nicknamed him "le Loup des Mers"--"the Sea Wolf" (23). In 1812, Cochrane proposed a plan to use "stink vessels" (ie, sulfur vessels) against the French Navy (23; 109). These ships, layered with coke and sulfur, would be sent amongst the French fleet and ignited to emit a choking gas (ie, a combination of sulfur dioxide, hydrogen sulfide, and carbonyl sulfide). The British Navy decided not to pursue his "secret plan"; however, he did communicate his plan with Lyon Playfair, who presented a modified proposal for gas warfare at the time of the Crimean War.
Crimean War (1853-1856). In 1853, at the beginning of the Crimean War, British scientist and politician Lyon Playfair (1818-1898) was appointed Secretary of the Department of Science, in which capacity he advocated the use of poison gas against the Russians:
|
At the beginning of the war I wrote a letter to the Prince Consort which he forwarded to the Master of the Ordnance, suggesting one or two applications of science to the purposes of war ... [A] proposal in my letter was to have a hollow brittle shell containing cyanide of cacodyl. This is an intensely poisonous substance, a few drops of which in a room would poison the occupants. Such a shell going between decks of a ship would render the atmosphere irrespirable, and poison the men if they remained at the guns. This suggestion was considered inadmissible by the military authorities, who stated that it would be as bad a mode of warfare as poisoning the wells of the enemy. There was no sense in this objection. It is considered a legitimate mode of warfare to fil shells with molten metal which scatters among the enemy, and produces the most frightful modes of death. Why a poisonous vapour which would kill men without suffering is to be considered illegitimate warfare is incomprehensible. War is destruction, and the more destructive it can be made with the least suffering the sooner will be ended that barbarous method of protecting national rights. No doubt in time chemistry will be used to lessen the suffering of combatants, and even of criminals condemned to death. Hanging is a relic of barbarism, because criminals might be put to death without physical torture (99). |
World War I. In the last century, poisonous warfare agents were used for the first time on the battlefields of Europe during World War I, which led to a rapid escalation in the development of gas masks (full-face respirator masks) and even early full-body hazmat suits (also referred to as haz-suits or hazardous materials suits), which required training to be used in combat situations and were soon employed in combat by soldiers on both sides in the war. The Germans, being the first to deploy chemical weapons at the second battle of Ypres (fought from April 22 to May 25, 1915), were the first to develop gas masks.
(Source: Deutsches Bundesarchiv [German Federal Archives]. Attribution: Bundesarchiv, Bild 183-F0313-0208-007 / CC-BY-SA 3.0. Creative Commons Attribution-Share Alike 3.0 Germany License, https://creativecommons.org/licenses/by...
(Source: War Department. Army War College. Historical Section. National Archives and Records Administration, cataloged under the National Archives Identifier (NAID) 55163709. This image is a work of a U.S. military or Departmen...
(Source: War Department. Army War College. Historical Section. National Archives and Records Administration, cataloged under the National Archives Identifier (NAID) 55162745. This image is a work of a U.S. military or Departmen...
(Source: War Department. Army War College. Historical Section. National Archives and Records Administration, cataloged under the National Archives Identifier (NAID) 55162641. This image is a work of a U.S. military or Departmen...
Men in the trenches with gas masks on ready to go over the top. Photographer: Charles Feinberg. National Archives and Records Administration, cataloged under the National Archives Identifier (NAID) 26423782. (This image is a wo...
United States Marines in training at Quantico, Virginia, going through drills in June 1918 "for the benefit of Secretary [Josephus] Daniels [Secretary of the Navy, 1862-1948] and other members of the government." Photographer: ...
"The war by fire has proved to be a boomerang to the Germans. ... The Allies are beginning to pay back the Germans in their own coin on the Western front, by means of certain devises [sic] to which, earlier in the war, the Germ...
In close combat in the presence of choking agents, one technique was to try to rip off the enemy soldier’s gas mask.
The use of chemical weapons also stimulated the development of simple but effective techniques to eliminate heavier-than-air toxic gases (eg, chlorine) from trenches, beating the gas from the trench by oscillating an Ayrton trench fan (a stiffened canvas blade on a stick). The fan was the idea of British engineer, mathematician, physicist, inventor, and suffragette Hertha Ayrton (born Phoebe Sarah Marks; 1854-1923).
U.S. Chemical Warfare Service drills, World War I (1918). (Source: War Department. Army War College. Historical Section. National Archives and Records Administration, cataloged under the National Archives Identifier (NAID) 5516...
From June 1917 to November 1918, the United States shipped nearly 4 million gas masks overseas along with nearly 2 million extra canisters, 350,000 horse gas masks, and approximately 28,000 trench fans (11).
In the setting of choking agents in World War I, an often-overlooked consideration was the need to protect both soldiers and horses.
World War I was a transition period between heavy reliance on horse-borne cavalry units and the later use of tanks. Horses were better than mechanized vehicles at traveling through deep mud and over rough terrain. Horses were used for reconnaissance, carrying messengers, hauling shells, and pulling artillery, ambulances, and supply wagons. France, not Germany, was the first to use a chemical weapon of sorts when they deployed tear gas in August 1914. The lachrymatory agent used was either xylyl bromide (smelling "pleasant and aromatic") or ethyl bromoacetate ("fruity and pungent"); both liquids must be atomized to be dispersed as weapons. These substances irritate the eyes, cause uncontrolled tearing, and, in large doses, can cause temporary blindness and breathing difficulties. Because symptoms usually resolve within 30 minutes, tear gas was never very effective as a weapon.
The use of lethal chemical weapons began on April 22, 1915, when, at 5 pm, German specialist troops released asphyxiating chlorine gas from cylinders embedded in the ground. A yellow-green cloud of chlorine gas drifted downwind and smothered the Allied line on the northern end of the Ypres salient, causing agony, panic, and death. Chlorine reacts with moisture to form hypochlorous acid (HClO) and hydrochloric acid (HCl); when this reaction occurs in the respiratory passages, the effect is devastating, causing acute and persistent damage (121; 52).
However, chlorine’s effectiveness as a weapon was short-lived because its color and odor made it easy to recognize, allowing time to don a gas mask. Even soldiers without gas masks could mitigate the adverse effects by placing water- or urine-soaked rags over their mouths and noses (the chlorine reacted with the water in the rag rather than water coating the respiratory passageways). Additionally, releasing the gas in a cloud posed problems, as the British learned when they first attempted to use chlorine at Loos, France, on September 25, 1915: the wind shifted, carrying the gas back onto their own men.
During World War I, the United States manufactured chemical weapons at U.S. military plants in Edgewood, Maryland; Stanford, Connecticut; Hastings-on-Hudson, New York; Kingsport, Tennessee; Croyland, Pennsylvania; and Willoughby, Ohio (11). The manufactured weapons included: (1) blister agents (ie, mustard gas and lewisite) and the precursor chemical sulfur monochloride (ie, disulfur dichloride); (2) choking or pulmonary agents (ie, bromobenzylcyanide, chlorine, chloripicrin, and phosgene); and (3) a vomiting agent (ie, diphenylchloroarsine, believed to penetrate the gas masks of the time and to cause violent sneezing, forcing removal of the protecting device; the Germans called it Maskenbrecher [mask breaker]) (11). In addition, five commercial chemical plants manufactured phosgene and mustard gas as well as precursor chemical-weapon chemicals: Oldbury Electro-Chemical Company in Niagra Falls, New York; Dow Chemical Compan in Midland Michigan; Frank Hemingway Inc. in Bound Brook, New Jersey; Charleston Chemical Company in Charleston, West Virginia; and National Analine & Chemical Company in Buffalo, New York (11). Deploying these weapons safely and coping with similar weapons deployed by the enemy required proper equipment and advanced training.
World War I: medical illustration and anti-chemical-warfare propaganda.
A. Kirkpatrick Maxwell (1884-1975). In 1915, Scottish artist A. Kirkpatrick Maxwell was asked by English surgeon Sir George Makens (1853-1933) to go to Boulogne, France, to create some surgical illustrations (34). Maxwell arrived in France just before the second battle of Ypres (fought from April 22 to May 25, 1915), at which the Germans used poison gas for the first time. Maxwell's drawings from Ypres came to the attention of British physician and physiologist Colonel Thomas Renton Elliott (1877-1961), who was then a medical consultant to the British Expeditionary Force in France. Elliott suggested to the newly formed Medical Research Council that Maxwell should be enlisted in the army as a medical illustrator, and on that recommendation, Maxwell was given the rank of Sergeant in the Royal Army Medical Corps and a room in the Medical Research Council’s laboratory in the 13th General Hospital in the Casino at Boulogne. Throughout World War I, Maxwell made over 1000 surgical illustrations of the results of gas attacks, war injuries, and post-mortem specimens, many of which were published in the British Journal of Surgery. The original illustrations were kept at the Royal College of Surgeons of England but were destroyed during the Blitz.
Among Maxwell's preserved illustrations is the face of a soldier suffering from the effects of phosgene gas poisoning during World War I and the contrasting pathological effects and microscopic appearance of pulmonary tissue from phosgene and sulfur mustard poisonings.
Pastel by Scottish artist A. Kirkpatrick Maxwell (1884-1975), ca. 1915. (Source: Wellcome Collection, London, UK. Wellcome Library no. 571751i. Creative Commons Attribution 4.0 International [CC BY 4.0] license, creativecommons...
Color halftone printed in two colors after Scottish artist A. Kirkpatrick Maxwell (1884-1975), ca. 1917. The victim died 19 hours after exposure. Most of the pulmonary alveoli are filled with edema fluid, and the walls of the a...
Color halftone printed in two colors after Scottish artist A. Kirkpatrick Maxwell (1884-1975), ca. 1917. The victim died at the end of the second day (40 hours after exposure). The bronchiole is filled with fibrin and pus cells...
Louis Raemaekers (1869-1956). Louis Raemaekers was a Dutch painter, caricaturist, and editorial cartoonist for the Amsterdam newspaper De Telegraaf during World War I.
After the Germans invaded Belgium, Raemaekers became a fierce critic of Germany and a proponent of Dutch opposition to German hostilities. His graphic cartoons depicted the German military in Belgium and portrayed the Germans as apes or barbarians and German emperor Kaiser Wilhelm II (1859-1941) as an ally of Satan. He was heavily criticized for endangering Dutch neutrality. The Dutch government repeatedly confiscated his work and pressured Raemaekers, his editor, and the owner of the paper to avoid provoking the Germans. Raemaekers eventually gave up his hopeless campaign to get the Netherlands to abandon its neutral stance. He left for London in November 1915, where his work received much acclaim. In early 1916, he signed a contract with the Daily Mail where his cartoons appeared regularly for the next 2 years.
Soon after his arrival in London, Raemaekers was contacted by Britain's War Propaganda Bureau, Wellington House, to ensure mass distribution of his anti-German artwork in England and elsewhere. This ushered in his important role in Allied war propaganda. Forty of his cartoons were published in Raemaekers' Cartoons (1916), which was promptly translated into 18 languages and distributed in neutral countries around the world. Multiple other books with his propaganda cartoons soon followed.
At the request of Wellington House, Raemaekers visited the United States in 1917 to draw attention to his work and help promote American support for involvement in the war as well as anti-German sentiment. His timing was auspicious because the United States had declared war on Germany shortly before. His tour was a triumph; Raemaekers gave lectures and interviews, was a popular guest at society functions, and met President Woodrow Wilson (1856-1924) and former President Theodore Roosevelt (1858-1919). By October 1917, more than 2000 American newspapers had published Raemaekers’ cartoons in millions of copies.
Among Raemaekers’ arresting images are depictions of a poisonous gas attack on the Canadians in Flanders on April 24, 1915, and the desperate efforts of hospitalized a soldier to breathe after a chemical weapons attack, with the sarcastic title, "Gassed! -- another victory for KULTUR," conveying its propagandistic intent. Other more abstract and clearly propagandistic images included Raemaekers' (1) "The Hun Ministry of Munitions," portraying the personification of Germany amidst stocks for chemical and germ warfare (the personification is leaning his right forearm on a tank labeled "Poison Gas"); (2) a large snake, symbolic of a gas attack, striking at a sleeping soldier; and (3) "— a toast to Kultur" in which a skeletal figure is shown drinking a glass of toxic red chemicals, representing the death of civilization by chemical warfare.
Colored chalks by Dutch painter, caricaturist, and editorial cartoonist Louis Raemaekers (1869-1956), ca. 1918. The print of this composition was "Drawn specially for the Graphic"; it was published in the Graphic on August 3, 1...
Colored chalk by Dutch painter, caricaturist and editorial cartoonist Louis Raemakers (1869-1956), 1916. (Source: Wellcome Collection, London, UK. Wellcome Library no. 46015i (Photo number: V0018240). Creative Commons Attributi...
After a crayon drawing by Dutch painter, caricaturist, and editorial cartoonist Louis Raemakers (1869-1956). (Source: Wellcome Collection, London, UK. Wellcome Library no. 46015i (Photo number: V0018240). Creative Commons Attri...
Treaty of Versailles (1919). In the Treaty of Versailles (1919) at the end of World War I, the victorious Allies reaffirmed a prohibition of the use of poisonous gases in warfare and forbade Germany to manufacture or import them. Similar provisions were included in the peace treaties with Austria, Bulgaria, and Hungary.
Geneva Protocol (1925). The 1925 Geneva Protocol prohibited the use of chemical and biological weapons in war. The Protocol was drafted and signed at a conference held in Geneva under the auspices of the League of Nations from May 4 to June 17, and it entered into force on February 8, 1928. It specifically precluded "the use in war of asphyxiating, poisonous, or other gases and of all analogous liquids, materials, or devices." Despite this hopeful development, countries continued to develop chemical warfare capabilities, and some countries considered chemical warfare a suitable means to achieve military goals.
Germany's development of nerve agents (1936-1949). Nerve agents were discovered inadvertently by German chemist Gerhard Schrader (1903-1990) at the IG Farben chemical conglomerate. Schrader had been charged with developing new insecticides to help reduce Germany’s reliance on food imported from abroad and had been successful in discovering several very effective insecticides, including "bladan" (the first fully synthetic contact insecticide, containing the organophosphate compound hexaethyl tetraphosphate), and parathion (E 605). After failing to make viable fluorine- and sulfur-based pesticides, Schrader began experimenting with molecules that combined phosphorus and cyanide. An early candidate was so poisonous that Schrader was hospitalized for several weeks after being exposed to trace amounts. On December 23, 1936, Schrader synthesized "preparation 9/91," later named tabun; dilute solutions destroyed insect food pests but also caused dyspnea, vomiting, diarrhea, mydriasis, sialorrhea, diaphoresis, and death in apes and other mammals.
Although this indiscriminate poison was clearly not a safe pesticide, IG Farben alerted the German military about this new poisonous compound. During World War II, teams led by Schrader discovered two more organophosphate nerve agents and then a fourth after the war, yielding the following: tabun (1936), sarin (1938), soman (1944), and cyclosarin (1949). Nazi Germany manufactured an extensive stockpile of military-grade nerve agents but never used them against the Allies. The Nazi nerve agent stockpiles were only discovered at the end of World War II.
Battle of Shanghai 1937. The Battle of Shanghai was the first of the 22 major engagements fought between the National Revolutionary Army of the Republic of China and the Imperial Japanese Army of the Empire of Japan at the beginning of the Second Sino-Japanese War. It was one of the largest and bloodiest battles of the entire war and is often regarded as the battle where World War II started. As a precaution, Japanese Special Naval Landing Forces wore gas masks in the event the Chinese would resort to chemical weapons.
The Battle of Shanghai was the first of the 22 major engagements fought between the National Revolutionary Army of the Republic of China and the Imperial Japanese Army of the Empire of Japan at the beginning of the Second Sino-...
World War II and the Holocaust. Unlike World War I, World War II did not see battlefield use of chemical weapons, even though that possibility was a worrying concern--so much so, in fact, that British Prime Minister Winston Churchill carried a gas mask with him. In addition, the British government issued gas masks to its citizens along with directions on the use of a respirator in case of a chemical weapon attack and developed poison gas identification kits that were issued to civil defense personnel, usually ARP (Air Raid Precaution) wardens. The kits contained eight small, labeled glass bottles and some sachets of chemicals in a leatherette case.
(Source: Chart of war gases. Issued by the Ministry of Home Security. London: H.M. Stationary Office, 1942. Courtesy of the Wellcome Library, London, UK. Reference number: b15316890. Public domain.)
During World War II, Nazi Germany and its collaborators conducted widescale poisoning and genocidal extermination of European Jews. Between 1941 and 1945, Nazi Germany and its collaborators systematically murdered some 6 million Jews across German-occupied Europe--around two thirds of Europe's Jewish population. The murders were carried out in mass shootings, by a policy of extermination through labor and starvation in concentration camps, and in gas chambers and gas vans in German extermination camps, especially in occupied Poland (ie, Auschwitz-Birkenau, Bełżec, Chełmno, Majdanek, Sobibór, and Treblinka). The gas chambers consisted of a sealed chamber into which a poisonous or asphyxiant gas was introduced, usually carbon monoxide or hydrogen cyanide (Zyklon B), both of which specifically inhibit cytochrome c oxidase of the human mitochondrial respiratory chain (03; 24). The gas vans or gas wagons (German: Gaswagen) were simply trucks re-equipped as mobile gas chambers. Nazi Germany developed and used gas vans on a large scale as an extermination method to murder inmates of asylums, Jews, Poles, Romani people, and prisoners in occupied Poland, Belarus, Yugoslavia, and the Soviet Union.
Zyklon B was the trade name of a cyanide-based pesticide invented in Germany in the early 1920s. Hydrogen cyanide was first used as a pesticide in California in the 1880s for fumigating citrus trees. Research by German chemist Fritz Haber (1868-1934) of the Kaiser Wilhelm Institute for Physical Chemistry and Electrochemistry led to the founding in 1919 of Deutsche Gesellschaft für Schädlingsbekämpfung mbH (Degesch), a state-controlled consortium formed to investigate military use of the chemical. Research at Degesch led to the development of Zyklon (ie, Cyclone in English, later known as Zyklon A), a pesticide that released hydrogen cyanide on exposure to water and heat. Degussa purchased Degesch in 1922; their chemists, including Walter Heerdt (1888-1958) and Bruno Tesch (1890-1946), devised a method of packaging hydrogen cyanide (prussic acid) in sealed canisters along with a cautionary eye irritant and an adsorbent (eg, diatomaceous earth). The new product, known as Zyklon B to distinguish it from the earlier version, was initially used to delouse clothing and fumigate ships, warehouses, and trains. The Nazis started using Zyklon B in extermination camps in early 1942 to murder prisoners during the Holocaust. Following the end of World War II, Tesch was arrested by the British as a war criminal, tried, and executed in 1946 for knowingly selling the product for use on humans.
After World War II. After World War II, new types of organophosphorus chemical warfare agents were developed, such as Novichoks in the Soviet Union/Russia.
Iraq's use of chemical weapons against Iran in the Iran-Iraq War (1980-1988). Iraq used the blister agent mustard gas from 1983 and the nerve gas Tabun from 1985 as it faced attacks from Iranian troops and poorly trained but loyal volunteers.
In a retrospective study with a 39-year follow-up, chemical warfare survivors had heightened risks of respiratory disorders, liver cancer, and multiple myeloma (04).
Halabja massacre (1988). The Halabja massacre was a massacre of Kurdish people in Halabja in northern Iraq on March 16, 1988, during the closing days of the Iran-Iraq War. Iraqi war planes and artillery pounded Halabja with mustard gas and the nerve agents sarin and tabun, apparently as punishment because some Kurdish guerrilla forces had joined the Iranian offensive (50). The incident was the largest chemical weapons attack directed against a civilian-populated area in history, killing between 3200 and 5000 people and injuring 7000 to 10,000 more, most of them civilians.
1st Lt. Matthew Chau, commander of Border Team 3, 25th Infantry Division, patrols Halabja, Iraq. Buried in the village cemetery are many victims of the 1988 chemical weapons attack, ordered by Saddam Hussein. Photograph by Sgt....
In a retrospective case-control study of 230 survivors and 240 nonexposed control participants matched by age, gender, and occupation, females were more prevalent among survivors, and surviving females had less severe consequences of exposure than males (80). Most of the survivors (60%) had been exposed by multiple routes, and the respiratory system was the most common single exposure route. The vast majority (89%) of survivors had impaired activities of daily living. Respiratory and cardiac diseases were significantly more common in the survivors compared to the controls. Survivors with multiple chemical weapons exposure routes had significantly higher rates of activities of daily living impairment and cardiac disease, respiratory diseases, and miscarriage than those with a single exposure route.
Aum Shinrikyo chemical (and biological) terrorism. Between 1990 and 1995, the Aum Shinrikyo cult in Japan launched 17 known attacks with chemical and biological weapons, 10 with chemical weapons (four with sarin, four with VX, one with phosgene, and one with hydrogen cyanide) and seven with biological agents (four with anthrax and three with botulinum toxin, although these all failed because the microbial strains were nonvirulent). In addition, cult members killed 20 dissident members with VX. The cult has also been linked more tenuously to more than 19 other chemical and biological weapon attacks and attempted attacks (13 attacks where Aum involvement is suspected and six possible copycats).
Tokyo subway sarin attack (1995). On June 27, 1994, in a dry run for a subsequent and more massive act of domestic terrorism, the Aum Shinrikyo cult released sarin from a vehicle driving through the industrial and resort city of Matsumoto, Japan, killing seven people and injuring some 500 more.
The cult perpetrated a major act of domestic terrorism in Tokyo on March 20, 1995. In five coordinated attacks, the perpetrators released sarin on three lines of the Tokyo Metro (then the Teito Rapid Transit Authority) during rush hour. The perpetrators had boarded the subway trains with plastic bags containing sarin and released the gas by simply poking holes in the bags with the metal tips of umbrellas. Even though emergency personnel responded quickly to the scene, the attack killed 13 people and injured over 1000, of whom 17 were critically injured (ie, requiring intensive care), 37 were moderately to severely injured (ie, with muscular twitching and gastrointestinal problems), and 984 were slightly injured (ie, with pinpoint pupils but no other symptoms).
Police, fire, and emergency medical personnel were very quickly on the scene in force. The Japan Defense Force Chemical troops were there within a couple of hours of being notified that they were needed. (Source: United States ...
When police later investigated documents seized in the cult office, they discovered the five-step process the cult used to manufacture sarin: (1) phosphorus trichloride was reacted with methanol to produce trimethyl phosphite; (2) trimethyl phosphite was converted to dimethyl methylphosphonate (DMMP) through a heat-induced rearrangement; (3) DMMP was reacted with phosphorus pentachloride by applying heat to produce methylphosphonyl dichloride; (4) methylphosphonyl dichloride was reacted with sodium fluoride to produce methylphosphonyl difluoride; and (5) methylphosphonyl difluoride and methylphosphonyl dichloride were mixed with isopropyl alcohol to produce sarin.
Tokyo subway attempted cyanide attacks (1995). On May 5, 1995, a burning paper bag was discovered in a toilet in Tokyo's Shinjuku subway station. On examination, it was found to be a hydrogen cyanide device; had it not been extinguished in time, it would have released enough gas into the ventilation system to potentially kill 10,000 commuters. Two months later, on July 4th, several undetonated cyanide devices were found at other locations in the Tokyo subway. Later, when police raided the facilities of the cult, stockpiles of chemicals were found that could be used to produce enough sarin to kill 4 million people.
Chemical Weapons Convention (1997). The Eighteen Nation Committee on Disarmament, sponsored by the United Nations in 1961, originally considered disarmament and nuclear test controls. Between 1965 and 1968, the committee negotiated the Treaty on the Non-Proliferation of Nuclear Weapons. By 1984, the Eighteen Nation Committee on Disarmament evolved into the Conference on Disarmament. On September 3, 1992, the Conference on Disarmament submitted to the United Nations General Assembly its annual report, which contained the text of the Chemical Weapons Convention (CWC)--officially the Convention on the Prohibition of the Development, Production, Stockpiling, and Use of Chemical Weapons and on their Destruction. The General Assembly approved the CWC on November 30, 1992, and the U.N. Secretary-General opened the CWC for signature in Paris on January 13, 1993. The CWC remained open for signature until it entered into force on April 29, 1997.
The CWC is an arms control treaty administered by the Organisation for the Prohibition of Chemical Weapons (OPCW, an intergovernmental organization based in The Hague, The Netherlands).
The CWC prohibits the large-scale use, development, production, and stockpiling of chemical weapons and their precursors, except in small amounts for limited purposes (ie, research, medical, pharmaceutical, or protective). The CWC augments the Geneva Protocol of 1925, which banned the use but not the development or possession of chemical and biological weapons. The CWC also includes extensive verification measures, such as on-site inspections, unlike the 1975 Biological Weapons Convention, which lacked a verification process.
As of October 2022, 193 countries have become parties to the CWC and accept its obligations. Members must follow this prohibition and destroy all current chemical weapons under OPCW verification. The CWC has provisions for systematic evaluation of chemical production facilities and for investigations of allegations of chemical weapons use and production based on other parties' intelligence. Israel signed the CWC in 1993 but has not yet ratified the agreement, whereas three other United Nations member states (Egypt, North Korea, and South Sudan) have neither signed nor acceded to the treaty.
Under the CWC, as of October 2022, 99% of the world's declared chemical weapons stockpiles have been destroyed (https://www.opcw.org, Accessed October 21, 2022).
Syrian Civil War (2012-2018). Chemical weapons were used repeatedly in the Syrian Civil War beginning in 2012. Both the Syrian government of Bashar al-Assad and militants of the Islamic State of Iraq and the Levant (ISIL), an Islamist terrorist organization, used chemical weapons, with most attacks carried out by the Syrian government. Chlorine was the most used agent, and sarin and sulfur mustard were also used (104). Chlorine was used in 92% of confirmed chemical weapons attacks attributable to the Assad regime (which was responsible for 98% of all recorded attacks over the course of the war), including the fateful April 7, 2018, attack on Douma by the Syrian army, which killed between 40 and 50 people and injured over 100 (104). Almost half of the attacks between 2014 and 2018 were delivered by aircraft, and less than a quarter were delivered from the ground. The deadliest attacks were the August 2013 sarin attack in Ghouta (reportedly killing between 281 and 1,729 people) and the April 2017 sarin attack in Khan Shaykhun (killing at least 89 people).
Attacks in 2013 prompted international pressure, resulting in an agreement with the Syrian Armed Forces to the supervised destruction of their chemical weapons. However, despite the disarmament process, completed on June 23, 2014, dozens of incidents with suspected use of chemical weapons followed throughout Syria. In April 2018, following at least 18 visits to Syria for inspections, the technical secretariat of the OPCW was unable to "verify that Syria had submitted a declaration that could be considered accurate and complete" (100).
Continued threat of chemical weapons. Despite the CWC, there is still a real threat of chemical weapon use (65), particularly in acts of terrorism and assassination. Consequently, soldiers routinely receive training in warfare during a chemical attack, particularly in using protective equipment during training with lachrymatory agents, realistic combat simulations, and the management of chemical weapons casualties.
Military trainees don their chemical warfare gear prior to entering the Nuclear, Biological, Chemical, Conventional Training Facility at Lackland Air Force Base, San Antonio, on September 16, 2002. The NBCC is part of week five...
Cpl. Brandon Dockery, a chemical and biological warfare defense instructor, fans tear gas in the gas chamber on November 27, 2013, on Parris Island, S.C., as recruits of Mike Company, 3rd Recruit Training Battalion, prove their...
Recruits of Charlie Company, 1st Recruit Training Battalion, shake their heads during chemical warfare defense training June 3, 2014, on Parris Island, S.C. The recruits shook their heads and performed other exercises to prove ...
Soldier in full NBC (nuclear biological and chemical) equipment is taking cover in a foxhole during a live-firing exercise prior to the start of operations in Kuwait. (This image is a work of a U.S. military or Department of De...
United States soldiers with the 44th Chemical Company, 2nd Chemical Battalion, 48th Chemical Brigade, based at Fort Hood, Texas, prepare to enter and clear a building of opposing forces during a simulated chemical attack at For...
Troops in NBC (nuclear biological and chemical) outfits with a “wounded” soldier on a stretcher during an exercise before the start of operations in Kuwait. (This image is a work of a U.S. military or Department of Defense empl...
United States Navy photo by Hospital Corpsman 3rd Class Raolito M Pambid, released. (This image was released by the United States Navy with the ID 030626-N- 0000R-001. This file is a work of a sailor or employee of the U.S. Nav...
In a descriptive analysis of the use of chemical, biological, radiological, and nuclear weapons by violent nonstate actors around the world from 1990 to 2020, 565 events were recorded; 89% involved single agents, and 11% involved multiple agents (111). There were 7,540 injuries (17.37 injuries per attack) for chemical agents, 59 (0.48 per attack) for biological agents, 50 for radiological events, and none for nuclear attacks. There were 965 fatalities (2.22/attack) from chemical agents, 19 (0.15/attack) from biological agents, and none from radiological and nuclear events. The United States (118), Russia (49), and Iraq (43) accounted for the top three countries where these events occurred.
Assassination weapons and weapons of chemical terrorism. VX and Novichok agents have been used to carry out political assassinations by both states and transnational terrorist organizations, as in the case of Russian dissident and opposition leader Alexei Navalny, who was poisoned in August 2020, evacuated in a comatose condition to Germany, and successfully treated at the Charite Hospital in Berlin.
In 12 attempted assassinations from 1994 to 2020, there were 12 designated targets, but one involved three simultaneous targets, and at least two individuals were targeted twice (Table 1) (Taro Takimoto, Noboru Mizonu, and possibly Emilian Gebrev) (112). Only three of the attempts killed the intended targets, whereas an additional 10 people (ie, not targets) were killed. Therefore, nerve agents are a low-efficiency assassination weapon with a high likelihood of collateral damage. A single attempt on three simultaneous targets injured more than 500 people who were apparently not the primary intended target (although that event in Matsumoto Japan in 1994 may have had a secondary terroristic element).
|
Date |
Perpetrator |
Agent |
Method |
Target |
Place |
Outcome |
|
June 27, 1994 |
Aum Shinrikyo |
Sarin (GB) |
Released from a converted truck |
Three judges who were to rule in a land dispute |
Japan (residential neighborhood, Matsumoto city) |
The judges survived, but the drifting cloud killed eight people and injured more than 500. |
|
Autumn 1994 |
Aum Shinrikyo |
VX |
Applied on car door handle |
Taro Takimoto (lawyer for Aum victims) |
Japan |
Unsuccessful |
|
Autumn 1994 |
Aum Shinrikyo |
VX |
Inserted into a keyhole |
Taro Takimoto (lawyer for Aum victims) |
Japan |
Unsuccessful |
|
November 28, 1994 |
Aum Shinrikyo |
VX |
Squirted from syringe onto target |
Noboru Mizonu (offered shelter to former Aum members) |
Japan |
Unsuccessful |
|
December 2, 1994 |
Aum Shinrikyo |
VX |
Squirted from syringe onto target |
Noboru Mizonu (offered shelter to former Aum members) |
Japan |
Unsuccessful but hospitalized for 45 days |
|
December 12, 1994 |
Aum Shinrikyo |
VX |
Injected from syringe |
Tadahito Hamaguchi (misidentified as police spy) |
Japan |
Killed |
|
January 4, 1995 |
Aum Shinrikyo |
VX |
Squirted from syringe onto target |
Hiroyuki Nagaoka (head of the Aum Victims Society) |
Japan |
Unsuccessful; hospitalized for several weeks |
|
August 1, 1995 |
Russia?1 business partner? |
Novichok agent |
Applied on telephone |
Ivan Kivelidi (founder of Rosbiznesbank) |
Russia (Moscow) |
Killed. His secretary, Zara Ismailova, was also killed. |
|
April 28, 2015 |
Russia |
Suspected Novichok agent |
? |
Emilian Gebrev (Bulgarian arms trader who had shipped weapons to Ukraine) |
Bulgaria |
Survived critical poisoning |
|
February 13, 2017 |
North Korea |
VX2 (binary VX)2 |
Sprayed on face |
Kim Jong-nam (exiled half-brother of Kim Jong-un, North Korean leader) |
Malasia (Kuala Lumpur International Airport) |
Killed |
|
March 4, 2018 |
Russia |
A-234 Novichok agent |
Applied to target's home door handle |
Sergei Skripal (former Soviet/Russian intelligence officer |
U.K. (Salisbury) |
Survived critical poisoning (as did his daughter Yulia and police detective Nick Bailey) |
|
June 30, 2018 |
Russia |
A-234 Novichok agent |
Inadvertent (recovered vial discarded by Russian agents) |
U.K. (Amesbury) |
Charlie Rowley killed; Dawn Sturgess survived critical poisoning | |
|
August 20, 2020 |
Russia |
Novichok agent |
? |
Alexei Navalny (Russian opposition politician) |
Russia (Tomsk Airport) |
Survived critical poisoning |
|
2 Binary chemical weapons utilize less toxic chemical precursors that form the intended nerve agent when combined. This improves the safety of storing, transporting, and disposing of the weapon. The VX2 binary agent had originally been developed by the United States, as had GB2 (binary sarin), whereas a binary Novichok agent had been developed by the Soviets. | ||||||
Chemical weapons of mass destruction. According to the U.S. Department of Homeland Security, "A weapon of mass destruction is a nuclear, radiological, chemical, biological, or other device that is intended to harm a large number of people" (Department of Homeland Security, Accessed October 16, 2022). This article addresses chemical agents used as weapons of mass destruction, as agents of chemical terrorism, and as assassination agents, providing a historical overview of chemical agents that have been developed or used in this capacity in the 20th century (Table 2) and then focuses on the class of chemical agents that selectively targets the nervous system: nerve agents.
|
Blood agents (cyanogenic agents) | ||
|
AC - hydrogen cyanide | ||
|
Choking agents (pulmonary agents) | ||
|
BBC - bromobenzyl cyanide | ||
|
Vesicants (blister agents) | ||
|
H - mustard gas (or sulfur mustard)3 | ||
|
Tear agents (lachrymatory agent; riot-control agents) | ||
|
CA - camite (bromobenzylcyanide) | ||
|
Vomiting agents | ||
|
CN-DM - combination of mace and the vomiting agent Adamsite5 | ||
|
Psycho agents | ||
|
BZ - 3-quinuclidinyl benzilate | ||
|
Nerve agents | ||
|
G series | ||
|
GA - tabun (EA1205) | ||
|
V series | ||
|
VE - VE nerve agent (EA1517) | ||
|
A series (Novichok agents)10 | ||
|
A-230 | ||
|
TZ - Saxitoxin11 | ||
|
Notes: 1 During World War II, cyanogen chloride's symbol was changed from CK to CC; when it became apparent that CC-marked munitions might be mistaken for CG (phosgene), the symbol was changed back. 2 Chloropicrin has the symbol PS, which was derived from the British town in which it was manufactured during World War I: Port Sunshine. 3 The original military designation for mustard gas was HS ("Hun Stuff"). Later, in England, HS signified mustard gas with about 25% solvent added to it, whereas in the United States, HS signified crude mustard. In World War II, the purity of mustard gas was improved through distillation, a form designated HD. When mustard gas was mixed with a thickener (Agent VV), it was given the symbol HV. Today mustard gas is indicated by the capital letter H, but HD is still commonly used. 4 The tear agent Mace, or Agent CN, had been formulated in several solvent forms, indicated by CNB (with benzene), CNC (with chloroform), and CNS (with chloropicrin and chloroform). 5 Mixtures of agents have been identified either with a hyphen (eg, CN-DM), combining letters of the two agents (eg, HD mixed with L is HL), or appending letters to the designation for a primary agent. 6 Numbers are occasionally added to military symbols to reflect particular preparations. With riot-control agents, a 1 signifies micro-pulverized (eg, CS1), and a 2 signifies microencapsulated (eg, CS2). 7 When the tear agent CS is formulated in a solvent, it is signified by CSX. 8 Binary chemical weapons are signified by adding a 2, as in binary sarin (ie, GB2). 9 When agents are thickened with the addition of a polymer, a T is usually added to the beginning of the symbol (eg, thickened soman is TGD). 10 A series of designations are the codenames assigned by Russia. 11 Agent TZ (saxitoxin) was derived from the last two letters of the surname of its principal investigator, biochemist Edward Shantz (1908-2005). Shantz worked for three decades in a Department of Defense laboratory at Fort Detrick, Maryland, before joining the University of Wisconsin in 1972. | ||
Scope of this article. For this article, agents that were not intended to cause direct harm to people are not addressed; therefore, for example, Agent Orange is not considered here because it was intended as a defoliant to eliminate forest cover and crops for North Vietnamese and Viet Cong troops. In addition to Agent Orange’s active ingredients, Agent Orange contained significant amounts of TCDD (2,3,7,8-tetrachlorodibenzo-p-dioxin), a type of dioxin; dioxin was not intentionally added to Agent Orange but was instead a byproduct produced during the manufacture of herbicides.
This article also does not address either the use of toxins (ie, an antigenic poison or venom of plant or animal origin that causes disease when present at low concentration in the body) or the use of nuclear or radiological agents.
Nerve agents. Nerve agents are acetylcholinesterase inhibitors that are used as poisons (12; 36). Nerve agents are mostly organophosphates, and sarin is the best-known nerve agent used for bioterrorism. These agents cause the rapid onset of a cholinergic crisis that can cause death within minutes but that is amenable to prompt treatment with specific antidotes (110). These agents induce nicotinic and muscarinic effects in the CNS and peripherally, causing paralysis of respiratory muscles and the medullary respiratory center, as well as bronchoconstriction and bronchorrhea (110).
Preparedness for nerve agent attacks is compromised by both a widespread tendency to downplay the presence of significant threat and a perception of low existing competency (115; 15). It is important for national security to provide education and training in managing nerve agents for frontline healthcare personnel as well as for medical students (115; 15).
|
• Chest tightness, bronchospasm, and difficulty breathing => apnea | |
|
• Miosis | |
|
• Dimness of vision | |
|
• Rhinorrhea | |
|
• Sialorrhea | |
|
• Bronchorrhea | |
|
• Diaphoresis | |
|
• Nausea, vomiting, abdominal cramps | |
|
• Urination (due to contraction of the detrusor muscles and relaxation of the internal sphincter muscle of the urethra) and defecation | |
|
• Twitching or jerking and muscle spasm | |
|
• Staggering | |
|
• Headache | |
|
• Drowsiness | |
|
• Confusion | |
|
• Convulsions | |
|
• Coma |
Cholinergic crisis is sometimes known by the mnemonic "SLUDGE syndrome": Salivation, Lacrimation, Urination, Defecation, Gastrointestinal distress, and Emesis.
Classic parasympathetic symptoms are emphasized by the mnemonic “DUMBBELS”: Defecation, Urination, Miosis, Bronchospasm, Emesis, Lacrimation, and Salivation.
The only available direct clinical experience with nerve agent poisoning that can be readily shared among most civilian clinicians comes from military training videos that the U.S. Army has released to the public showing the effects of battle simulations on animals.
In addition, there are clinical parallels with cholinergic crises due to some pharmaceuticals, organophosphate and carbamate insecticide poisoning (including accidental exposures and suicide attempts), and some forms of mushroom poisoning (ie, from species containing muscarine).
Nerve agents, such as sarin (GB) and tabun (GA), can be released into the air as a liquid spray (aerosol) or as a vapor. They can be absorbed into the body by inhalation, ingestion, skin contact, or eye contact, although ingestion is an uncommon route of exposure. Exposure to these agents can cause death within minutes.
A long-term study of Chinese subjects exposed to chemical warfare agents (a mixture of sulfur mustard and Lewisite) leaked from drums abandoned during World War II showed adverse sequelae, such as cognitive and emotional disorders and autonomic nervous system dysfunction (55).
Sarin exposure is associated with symptoms of organophosphate-induced delayed effects and chronic neurotoxicity. Delayed neurobehavioral and neurophysiological effects of acute sarin poisoning include impairment of both psychomotor performance and the vestibulocerebellar system. Distal sensory axonopathy has been reported as a late sequela of sarin intoxication. Sequelae of sarin poisoning, such as fatigue, asthenia, shoulder stiffness, and blurred vision, may persist for years after exposure.
Sarin and cyclosarin exposure during the 1991 Gulf War was associated with neurobehavioral dysfunction in war veterans on tasks involving fine psychomotor dexterity and visuospatial abilities 4 to 5 years after exposure (96). Brain pathology has been studied for 2 decades in veterans of the 1991 Persian Gulf War who were accidentally exposed to sarin in Iraq. MRI revealed reduced gray matter and white matter volumes, and neuropsychological testing showed errors of omission and a tendency to have slower responses (18). Sarin also causes immunotoxic effects and organophosphate-induced endocrine disruption.
|
• Sarin, an organophosphate, is a potent acetylcholinesterase inhibitor and causes toxic cholinergic hyperactivity in peripheral and central nervous systems. |
Acetylcholinesterase and cholinesterase inhibitors. Acetylcholinesterase is an enzyme that catalyzes the breakdown of the neurotransmitter acetylcholine to acetate and choline.
Physostigmine. The first identified acetylcholinesterase inhibitor was physostigmine, and the knowledge obtained from the study of physostigmine and related compounds greatly accelerated and supported the understanding of nerve agents. Physostigmine (also known as eserine from éséré, the West African name for the Calabar bean) is a highly toxic reversible cholinesterase inhibitor that occurs naturally in the Calabar bean and the fruit of the Manchineel tree. The properties of the Calabar bean and its potential medicinal uses attracted a great deal of interest in the mid-19th century and well into the 20th century (49; 120; 54; 83; 84; 97).
Physostigma venenosum, the Calabar bean or ordeal bean, is a leguminous plant that is endemic to tropical Africa, with a seed poisonous to humans. The Calabar bean contains physostigmine, an alkaloid that is a reversib...
The people of Old Calabar (also referred to as Callabar, Calabari, Calbari, and Kalabar) in Nigeria used Calabar beans or '"E-ser-e" as an ordeal poison administered to persons accused of witchcraft or other crimes: it was thought to affect only the guilty, so if a person accused of a crime ingested the beans without dying, they were considered innocent (26; 05; 84).
Physostigmine was "discovered" in 1846 by Scottish toxicologist and physician Sir Robert Christison, 1st Baronet (1797-1882), who undertook experiments on himself, such as taking large doses of Calabar bean (ie, containing physostigmine) (20). Christison proposed that physostigmine be used in the United Kingdom for humane execution of the condemned.
Sir Robert Christison became an authority on poisons, and his treatise on them was published in 1829. In the course of his research he undertook experiments on himself, such as taking large doses of Calabar bean (physostigmine)...
In 1862, Scottish physician Thomas Richard Fraser wrote his MD thesis on the Calabar bean: On the characters, actions, and therapeutic uses of the ordeal bean of Calabar (Physostigma venenosum) (39; 40; 41; 97; 33). Physostigmine as an antidote to atropine poisoning was described by a German ophthalmologist, Kleinwachter, in 1864, while working as the Second ophthalmologist at the Kaiser Hospital in Prague (63; 64; 84). Atropine as an antidote to physostigmine was reported by Fraser in 1870 and expanded on in 1871 and 1872 (42; 43; 44). Fraser recognized that there was "an antagonism" between the actions of these two drugs.
In 1863, Scottish ophthalmologist and surgeon Douglas Argyll Robertson (1837-1909) and English physician John William Ogle (1824-1905) introduced physostigmine into ophthalmological practice (87; 101).
Physostigmine as an antidote to curare poisoning was recognized by a Viennese physician, Pal, in 1900 (90; 91; 83).
Scottish neurologist Mary Broadfoot Walker (1888-1974) first demonstrated the effectiveness of physostigmine in the treatment of myasthenia gravis and, in so doing, established the first effective treatment for this disease (116; 117; 118; 119; 06; 32; 69; 95; 70; 61; 62; 57; 58; 94; 72; 86; 68; 14; 74).
In 1934, while working at St. Alfege’s Hospital in southeast London, Walker discovered that subcutaneous injection of physostigmine could temporarily reverse the muscle weakness associated with myasthenia gravis. She noted that the symptoms and signs of myasthenia were like those found in curare poisoning and that physostigmine was known to be an antidote to curare poisoning. Walker's findings were soon confirmed and also extended to neostigmine ("Prostigmin").
In 1935, American research chemist Percy Lavon Julian (1899-1979) and his longtime collaborator, Austrian chemist Josef Pikl, completed the total synthesis of physostigmine, which was previously only available from its natural source (60).
Organophosphate nerve agents. Organophosphate nerve agents are usually irreversible acetylcholinesterase inhibitors that precipitate a cholinergic crisis, with excess activation of muscarinic and nicotinic receptors when delivered to a target.
There are now three chemical series of nerve agents: the G, V, and A series.
Physostigma venenosum, the Calabar bean or ordeal bean, is a leguminous plant that is endemic to tropical Africa, with a seed poisonous to humans. The Calabar bean contains physostigmine, an alkaloid that is a reversib...
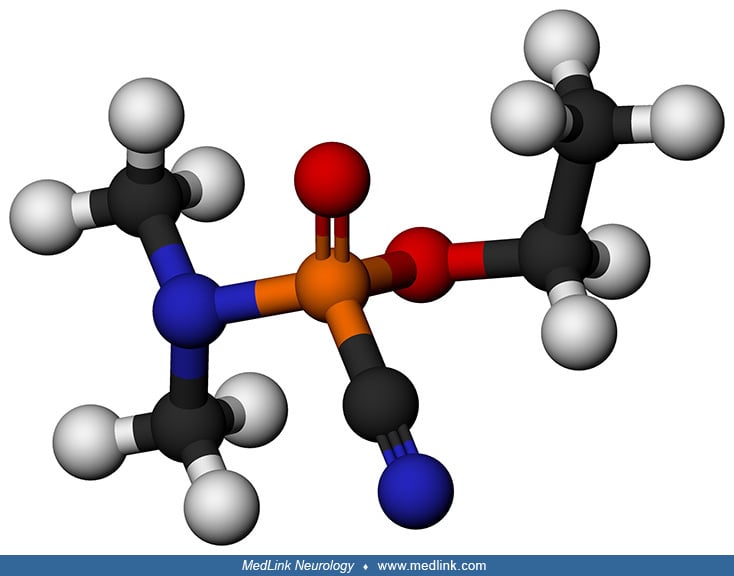
Sarin (GB). Sarin (O-isopropyl methylphosphonofluoridate) is a clear, colorless liquid that is odorless and tasteless yet highly neurotoxic in both its liquid and vapor states. Sarin is a potent acetylcholinesterase inhibitor and causes severe signs of toxic cholinergic hyperactivity in both the peripheral and central nervous systems. Cholinergic control of GABA release in the hippocampus can account for the occurrence of seizures in intoxicated subjects.
Animal studies suggest a central mechanism for respiratory paralysis in sarin neurotoxicity. A study of neuronal cells exposed to repeated sarin poisoning shows that ETS2 gene expression increases and affects the neurodegenerative pathway (89).
Tabun (GA). Tabun is a clear, colorless to pale, or dark amber liquid, reportedly with a fruity odor reminiscent of bitter almonds.
Soman (GD). Soman is a clear, colorless liquid that gives off a colorless vapor; soman discolors with aging to a dark brown color. It reportedly has an odor like camphor or rotting fruit.
VX (O-ethyl S-[2(diisopropylamino)ethyl]methylphosphonothioate). VX is a clear, amber-colored, oily liquid. Because VX is relatively persistent where it is dispersed, it is considered an area denial weapon; it is not considered a vapor hazard due to its relative nonvolatility.
VX is easily absorbed through the skin. One of the deadliest chemical weapons, a trace of VX can kill within minutes. As little as one drop of VX on the skin can be fatal. Under the Chemical Weapons Convention, VX has been internationally banned since 1997, but it remains a possible assassination weapon and a terrorist threat. Exposed to the environment, it degrades in a matter of hours on a concrete surface.
VR (O-Isobutyl S-[2-(diethylamino)ethyl]methylphosphonothioate). VR is a structural isomer of VX and has the potential to be used as a sabotage or terrorist agent. Acute exposure to a lethal dose of VR has been shown to cause cholinergic hyperfunction, incapacitation, seizures, convulsions, cardiorespiratory depression, and death.
Novichok agents. Novichok agents were secretly developed in the Soviet Union between the 1970s and 1990s (38; 122).
Knowledge of Novichok agents comes primarily from testimony and memoirs of dissident Russian chemist Vil Sultanovich Mirzayanov, former Chief of the Department of Counteraction against Foreign Technical Intelligence at the Russian State Union Scientific Research Institute for Organic Chemistry and Technology (GosNIIOKhT or ГосНИИОХТ ин Cyrillic) (108; 77). Mirzayanov described the state of chemical weapon disarmament in Russia and detailed the initiation of a secret Soviet chemical weapons initiative to develop Novichok agents (Новичок in Russian, transliterated to Novichok, meaning “newcomer” agents). Mirzayanov had worked for 26 years at GosNIIOKhT and witnessed Pyotr Petrovich Kirpichev and his assistants developing this new series of nerve agents--the A series--derived from G-series and V-series agents.
The first compound of this new series to be synthesized and tested (in 1973) was a derivative of sarin given the codename A-230 and referred to as substance 84 in internal reports of GosNIIOKhT.
The first three of these (Substance-33, A-230, and A-232) were produced at a GosNIIOKhT facility in Russia using an organophosphate structural backbone. A-230 was five to eight times as toxic as VX (referred to as substance 33). A technical difficulty was that it crystalized when stored at -10 °C, which was solved by adding N,N-dimethylformamide. Kirpichev and his group synthesized and tested the derivatives A-232, A-234, A-242, and A-262. A-232 and A-234 were much more volatile than VX. A-242 and A-262 were the first solid neurotoxic agents synthesized. A-230 was tested in the 1980s and, in 1990, was approved by the Soviet Army for use in ammunition. Mass production was begun at a new chemical weapons factory in Pavlodar, Kazakhstan.
Sergei Viktorovich Skripal, a former Russian military intelligence officer who acted as a double agent for the United Kingdom’s intelligence services during the 1990s and early 2000s, was poisoned along with his daughter with a Russian Novichok nerve agent in Salisbury, England, on March 4, 2018. Two Russian agents identified on CCTV cameras in Salisbury at the time of the attack were charged with attempted murder. On August 20, 2020, Russian opposition leader and anti-corruption activist Alexei Navalny was similarly poisoned with a Novichok nerve agent during a flight and was hospitalized in serious condition. He was evacuated to the Charité hospital in Berlin, Germany, 2 days later. The use of a Novichok nerve agent was confirmed by five laboratories certified by the Organisation for the Prohibition of Chemical Weapons.
An epidemiological study of sarin poisoning was carried out in 1994 when several liters of sarin were released during the night by terrorists in Matsumoto City, Japan (81). The study involved approximately 500 affected people in a population of approximately 2000. The geographical distribution of sarin victims was closely related to the direction of the wind. There were no deaths. Three weeks after the intoxication, 129 victims still had some symptoms, such as dysesthesia of the extremities, and some still had symptoms a year after the intoxication.
At a political level, the Chemical Weapons Convention (CWC) is meant to help protect against the use of nerve agents, but, as has been repeatedly observed, the CWC does not preclude the use of these poisons by terrorists and autocratic regimes. The Organisation for the Prohibition of Chemical Weapons (OPCW) is charged with implementing the provisions of the Chemical Weapons Convention. Four United Nations states are not party to the CWC: Egypt, Israel, North Korea, and South Sudan.
At an individual level, for military combatants at risk of chemical weapons attacks, special gas masks are available and have been used to prevent toxicity by inhalation during threats of nerve gas attacks. After rapidly assessing that a nerve agent attack has occurred, emphasis must be placed on decontamination and protection from exposure of both rescuers and medical personnel.
A cholinergic crisis from overstimulation of nicotinic and muscarinic receptors is usually secondary to the inactivation or inhibition of acetylcholinesterase, the enzyme responsible for the degradation of acetylcholine.
Cholinergic crisis can be caused by prescription drugs during clinical use or with a drug overdose (88). Example drugs include those used to treat myasthenia gravis (eg, edrophonium, pyridostigmine, and neostigmine), glaucoma (ie, pilocarpine), and Alzheimer disease drugs (eg, rivastigmine and donepezil). Cholinergic crisis caused by pharmaceutical cholinesterase inhibitors is rare but potentially life-threatening (88). Clinical settings where this is more likely to occur include myasthenia gravis, dementia, benign prostatic hypertrophy, and neurogenic bladder. Patients who require mechanical ventilation and either catecholamines or atropine have a poorer prognosis (88).
Self-poisoning with organophosphate pesticides is a major health problem worldwide (56; 125; 01; 93; 53; 103; 73; 92), but homicidal (25) and accidental (123) poisoning is also reported. Usually, the route of exposure is ingestion, but other routes (eg, subcutaneous injection) occasionally occur (09). Organophosphorus poisoning is characterized by an acute cholinergic crisis resulting from the inhibition of acetylcholinesterase. Other manifestations are an intermediate neurotoxic syndrome and delayed polyneuropathy (106; 78; 30; 28; 29; 85; 105; 107; 124; 01; 08; 27; 19; 92). Uncommon manifestations include chronic organophosphate-induced neuropsychiatric disorders (76; 46), transient parkinsonism (07; 103), cerebellar dysfunction (37), a myasthenic-like syndrome (102), and rhabdomyolysis (45).
Mushroom poisoning with cholinergic toxicity results from the ingestion of muscarine-containing species in various genera (eg, Clitocybe and Inocybe). Cholinergic effects of abdominal cramping, diaphoresis, salivation, lacrimation, bronchospasm, bronchorrhea, and bradycardia usually occur within 30 minutes. The duration is dose-dependent but typically shorter than with other sources of cholinergic poisoning (eg, pesticide poisoning).
Sarin exposure can be confirmed by examining material at the scene of exposure or the patient's clothing. The Argonne National Laboratory in Lemont, Illinois, outside of Chicago, has developed a method of screening air and water samples for sarin using solid-phase microextraction-gas chromatography-mass spectrometry; it is ideal for collecting air and water samples in the field and transporting samples safely to the laboratory.
Blood chemistry shows decreased serum cholinesterase activity following exposure to sarin. Urine specimens of poisoned individuals typically have measurable quantities of various sarin metabolites, such as methylphosphonate fluoridate, diethyl methylphosphonate, and ethyl isopropyl.
Biomarkers of exposure to nerve agents, metabolites, and adducts to proteins are required for diagnosis to guide appropriate treatment in the event of an exposure and for monitoring the health of workers handling nerve agent samples in laboratories (10).
The standard treatment of organophosphate nerve agent poisoning combines an antimuscarinic drug (eg, atropine), an anticonvulsant drug (eg, diazepam), and cholinesterase reactivators, including different types of oximes.
The oximes hydrolytically cleave the organophosphates from acetylcholinesterase, restoring enzymatic function, which depends on the type of agent and the reactivator used. In practice, the primary treatment is with atropine to control muscarinic symptoms and with oximes to reactivate the cholinesterases and treat the nicotinic symptoms (13).
The U.S. Army uses pralidoxime to reactivate acetylcholinesterase inhibited by organophosphate nerve agents. Early administration of pralidoxime chloride maximizes antidotal efficacy, but it does not cross the blood-brain barrier at therapeutically relevant levels.
Although pyridinium oximes have been used to manage poisoning with organophosphorus compounds for several decades, a significant drawback is an inefficient restoration of brain acetylcholinesterase activity due to difficulty penetrating the blood-brain barrier (67). Uncharged reactivators with increased bioavailability in the central compartment are more promising for treating organophosphate poisoning. Chemical modifications, such as glycosylation of pyridine aldoximes, facilitate increased blood-brain barrier penetration via the GLUT-1 transport system (47; 75).
Benzodiazepines, such as diazepam, are the current anticonvulsants of choice for controlling nerve agent-induced life-threatening seizures, status epilepticus, and brain injury. However, benzodiazepines are less effective for delayed treatment of status epilepticus, which is characterized by rapid desensitization of synaptic GABAA receptors and benzodiazepine resistance (98).
Sarin. The standard treatment for sarin-like nerve agent exposure is the postexposure injection of atropine, a muscarinic receptor antagonist, accompanied by an oxime, an acetylcholinesterase reactivator, and diazepam (02). Atropine is recommended as the first-line treatment, and the dose should be titrated to dry secretions and resolve bronchoconstriction. The AtroPen (atropine autoinjector), approved by the U.S. Food and Drug Administration, is indicated as an initial treatment of the muscarinic symptoms of nerve agent poisonings. Each prefilled autoinjector provides a dose of the antidote atropine, an anticholinergic drug that reduces secretions in the mouth and respiratory passages, relieves the constriction and spasm of respiratory passages, and may reduce the paralysis of respiration resulting from actions of the toxic agent on the CNS. The effectiveness of atropine depends on the route of administration: intravenous injection is quicker than the autoinjector, and the slowest response is intramuscular injection with a conventional syringe.
Respiratory support is required in severe acute poisoning. Benzodiazepines, in addition to atropine, should be administered if seizures develop. Early and aggressive medical therapy is the key to preventing the morbidity and mortality associated with nerve agent poisoning.
Physostigmine is an effective treatment against sarin intoxication. Pyridostigmine can be used as a prophylactic agent but does not obviate the need for atropine use (48).
A combination of atropine (one of the recommended pyridinium oximes: pralidoxime, trimedoxime, obidoxime, and HI-6) and diazepam is used for the treatment of organophosphorus poisoning in humans (59). Additional treatments, such as magnesium sulfate, sodium bicarbonate, gacyclidine, benactyzine, tezampanel, hemoperfusion, antioxidants, and bioscavengers, have also been used for organophosphate nerve agent poisoning (79).
An experimental study of sarin exposure in mice showed that neuronal death is mostly due to necrosis and severe astrocytosis that evolves over a period of 24 hours following exposure, suggesting that the timing of possible treatments should be determined by early events following exposure (71).
Rapid administration of antidotes. A special injector called Antidote Treatment Nerve Agent Autoinjector is available to the U.S. Army. This multichambered autoinjector delivers two antidotes in a single injection while maintaining separation of the two drugs in the injector and at the injection site.
Although there were four pregnant women among those hospitalized with mild to moderate symptoms following the sarin attack in the Tokyo subway in 1995, there is no mention of any complications related to pregnancy. However, acute organophosphorus pesticide poisoning during pregnancy has been reported to lead to spontaneous abortion.
Experimental studies show no evidence of developmental toxicity in the rat or the rabbit following exposure to sarin during embryonic differentiation and major organogenesis, even at a dose that produced maternal toxicity.
All contributors' financial relationships have been reviewed and mitigated to ensure that this and every other article is free from commercial bias.

Douglas J Lanska MD MS MSPH
Dr. Lanska of the University of Wisconsin School of Medicine and Public Health has no relevant financial relationships to disclose.
See ProfileNearly 3,000 illustrations, including video clips of neurologic disorders.
Every article is reviewed by our esteemed Editorial Board for accuracy and currency.
Full spectrum of neurology in 1,200 comprehensive articles.
Listen to MedLink on the go with Audio versions of each article.
MedLink, LLC
3525 Del Mar Heights Rd, Ste 304
San Diego, CA 92130-2122
Toll Free (U.S. + Canada): 800-452-2400
US Number: +1-619-640-4660
Support: service@medlink.com
Editor: editor@medlink.com
ISSN: 2831-9125
Neuropharmacology & Neurotherapeutics
May. 14, 2026
Neuropharmacology & Neurotherapeutics
Apr. 23, 2026
Neuropharmacology & Neurotherapeutics
Apr. 20, 2026
Neuro-Ophthalmology & Neuro-Otology
Apr. 07, 2026
General Neurology
Apr. 06, 2026
Neuro-Ophthalmology & Neuro-Otology
Mar. 30, 2026
Neuro-Ophthalmology & Neuro-Otology
Mar. 30, 2026
Headache & Pain
Mar. 29, 2026